Dr. Francisco Gómez Veiga. CHUAC, A Coruña. Controversias en el screening del cáncer de próstata: A quién y cuándo debo hacer un PSA?
|
|
|
- Agustín Ramos Bustos
- hace 8 años
- Vistas:
Transcripción
1 CHUAC, A Coruña
2 CONTROVERSIAS EN EL SCREENING DEL CÁNCER DE PRÓSTATA: A QUIÉN Y CUÁNDO DEBO HACER UN PSA? Complexo Hospitalario Universitario de A Coruña
3 Introducción Discrimina el PSA Tumores y agresividad? CONTROVERSIAS EN EL SCREENING DEL CÁNCER DE PRÓSTATA: A QUIÉN Y CUÁNDO DEBO HACER UN PSA? Mejores Marcadores? Datos de despistaje? Pros y Cons del Despistaje? Cómo debemos estratificar Riesgos? Conclusiones
4 Center M. International variation in prostate cancer. Incidence and mortality rates. Eur Urol 2012; 61:
5 Jemal, Ca. Cancer J.Clin CA. PRÓSTATA - ESTADISTICAS R.P Sx bx PSA
6
7 Test de PSA? Riesgo Beneficio
8 Sensibilidad MARCADOR IDEAL Marcador Ideal SIMPLE DE OBTENER REPRODUCIBLE COSTE/EFECTIVO Especificidad
9 Introducción HBP & Cáncer de próstata El Dilema PSA
10 0
11 Progresión en el tiempo de las formas de PSA BPSA 10 30% Free PSA ppsa PSA Other ipsa* 70 90% PSA ACT Complex PSA PSA * ipsa= inactive PSA/not clipped/ not propsa ,5 4
12 PSA. Niveles Thompson IM, Pauler DK, Goodman PJ, Tangen CM, Lucia MS, Parnes HL, et al. Prevalence of prostate cancer among men with a prostate specific antigen level < or = 4.0 ng per milliliter. N Engl J Med. 2004; 350:
13 Relationship of total PSA, complexed PSA and % complexed PSA with the presence of tumor and tumor aggressiveness N=3098 biopsies Total PSA No Cancer (%) Cancer (%) Gleason <7 (%) Gleason 7 (%) Positive cores 2 (%) Relationship between total PSA, diagnosis of prostate cancer and tumor aggressiveness Positive cores >2 (%) TOTAL Gómez Veiga FG*, Ponce J, et al. Models to predict prostate cancer aggressiviness in patients candidates to biopsy with PSA J Urol 2011
14 Es esto suficiente? Qué factores influyen para que la biopsia pueda ser positiva, y si lo es que potencial de agresividad Puede tener? Edad, Volumen, PSA c, %C/T PSA, Tacto Rectal, Técnica de biopsia?
15 PSA y Detección PSA, ng/ml >10 DRE RESULTS BIOPSY % WITH CANCER BIOPSY % WITH CANCER BIOPSY % WITH CANCER BIOPSY % WITH CANCER Normal Indicative of cancer No Yes 1 5 No Yes Yes Yes Yes Yes >50 >75 Catalona WJ, et al. Use of the percentage of free prostate specific antigen to enhance differentiation of prostate cancer from bening prostatic disease. JAMA 1998; 279 (19):
16 Factores Efecto del volumen prostático y la DPSA y su relación con la agresividad PATIENT AND TUMOR CHARACTERISTICS IN RELATION TO PSA DENSITY PSAD (ng/ml/cc) < > 0.19 P Value Mean pt age ± SD 65 ± ± ± ± No. organ confined (%) 357 (82) 283 (75) 171 (75) 192 (55) <0.001 No. Gleason sum greater than 7 (%) 86 (20) 102 (27) 64 (28) 157 (45) <0.001 No. Tumor vol greater than 0.5 cc (%) 91 (21) 91 (25) 74 (33) 157 (56) <0.001 No. With progression (%) 26 (6) 31 (9) 22 (10) 67 (21) <0.001 Benson MC, et al. J Urol 1992; 147: PSA PSA En los pacientes con cáncer, la mayor agresividad histológica (Gleason >7) se corresponde con un volumen prostático menor que cuando el tumor es más indolente (33,5 ± 16,5 c.c. vs. 41,5 ± 21,6 c.c., p<0,001), relación que se también aplica si se considera la presencia o no de tejido tumoral en 2 o más cilindros de biopsia (40,3 ± 21,3 c.c. si 2 cilindros positivos, 34,9 ± 17,8c.c.si>2cilindros positivos, p<0,001).
17 PSA. Evolución. Velocidad 1 PSA. Cinética Detection of Life-Threatening Prostate Cancer With Prostate-Specifi c Antigen Velocity During a Window of CurabilityH. Ballentine Carter. J Natl Cancer Inst 2006;98: 1521 Men with PSA velocity above 0.35 ng/ml per year had a higher relative risk of prostate cancer death than men with PSA velocity of 0.35 ng/ml per year or less 0 2, PSA PSA. Evolución. Velocidad 2 0 2, PSA
18 PSA Edad? Comorbilidad?
19 Marcadores biológicos Calicreina humana 2 (hk2) Antígeno precoz de cáncer de próstaa (EPCA) Gen del cáncer de próstata 3 (PCA3) Genes de fusión TMPRSS2 ETS Factor de crecimiento β1(tgf β1) e interleucina 6 (IL6) Perfil proteómico microrna Glutation S transferasa (GSTP1) PSMA Marcadores Genéticos. SNP Cromogranin A (GRN A) 2 propsa
20 In contrast to the serum markers discussed previously, the new biomarker prostate cancer antigen 3 (PCA3) is measured in urine sediment obtained after prostatic massage. Determination of this PCa specific RNA is experimental. At a population level it appears to be helpful, but its impact at a single patient level remains highly questionable. Table 2 Guidelines for the diagnosis of prostate cancer Heidenreich A, et al. EAU Guidelines on prostate cancer. Part 1: screening, idagnosis, and treatment of clinically localised disease. Eur Urol, 2011;
21
22 Prostate cancer is the second most common cause of male cancer deaths in the United States. However, the full range of prostate cancer genomic alterations is incompletely characterized. Here we present the complete sequence of seven primary human prostate cancers and their paired normal counterparts. Several tumours contained complex chains of balanced (that is, copy neutral ) rearrangements that occurred within or adjacent to known cancer genes. Rearrangement breakpoints were enriched near open chromatin, androgen receptor and ERG DNA binding sites in the setting of the ETS gene fusion TMPRSS2 ERG, but inversely correlated with these regions in tumours lacking ETS fusions. This observation suggests a link between chromatin or transcriptional regulation and the genesis of genomic aberrations. Three tumours contained rearrangements that disrupted CADM2, and four harboured events disrupting either PTEN (unbalanced events), a prostate tumour suppressor, or MAGI2 (balanced events), a PTENinteracting protein not previously implicated in prostate tumorigenesis. Thus, genomic rearrangements may arise from transcriptional or chromatin aberrancies and engage prostate tumorigenic mechanisms. Science 2005; 310: Figure 4 Disruption of CADM2 and the PTEN pathway by rearrangements. a, Location of intragenic breakpoints in CADM2. b, CADM2 break apart demonstrated by FISH in an independent prostate tumour. c, Location of intragenic breakpoints in PTEN (top) and MAGI2 (bottom). d,magi2 inversion demonstrated by FISHin an independent prostate tumour, using probes flanking MAGI2 (red and green) and an external reference probe also on chromosome 7q (green). Berger MF, et al. The genomic complexity of primary human prostate cancer. Nature 2011; 470:
23 Genesis and Progression of Gene Fusions in Prostate Cancer Kumar Sinha C, et al. Recurrent gene fusions in prostate cancer. Nat Rev Cancer 2008; 8 (7):
24 Gamut of Gene Fusions in Prostate Cancer Kumar Sinha C, et al. Recurrent gene fusions in prostate cancer. Nat Rev Cancer 2008; 8 (7):
25 Bill-Axelson A, et al. Radical prostatectomy versus watchful waiting in early prostate cancer. N Engl J Med 2011; 364: Figure 2. Cumulative incidence of death from prostate cancer and development of metastases among men with lowrisk prostate cancer
26 Bill-Axelson A, et al. Radical prostatectomy versus watchful waiting in early prostate cancer. N Engl J Med. 2005; 352: Resultados a 5 y 10 años del seguimiento en pacientes con cáncer de próstata tratados con prostatectomía radical (PR) y esperar y ver (EV), en %
27 Bill-Axelson A, et al. Radical prostatectomy versus watchful waiting in early prostate cancer. N Engl J Med 2011; 364 (18): Figure 1. Cumulative Incidence of Death from Any Cause, Death from Prostate Cancer, and Development of Metastases. The cumulative incidence of death from any cause, death from prostate cancer, and development of metastases in the radical-prostatectomy group and the watchful-waiting group is shown in the total cohort (Panels A, B, and C), in men 65 years of age or older (Panels D, E, and F), and in men younger than 65 years of age (Panels G, H, and I).
28
29 Situación Schröder FH, et al. Prostate cancer mortality at 11 years of follow up. N Engl J Med. 2012; 366 (11): Schröder FH, et al. Screening and prostate cancer mortality in a randomized European study. N Engl J Med. 2009; 360:
30 Mortality results from the Göteborg randomised population-based prostate-cancer screening trial. Hugosson. Lancet Oncology 2011 Methods In December, 1994, men born between 1930 and 1944, randomly sampled from the population register, were randomised by computer in a 1:1 ratio to either a screening group invited for PSA testing every 2 years (n=10 000) or to a control group not invited (n=10 000). Men in the screening group were invited up to the upper age limit (median 69, range years) and only men with raised PSA concentrations were off ered additional tests such as digital rectal examination and prostate biopsies. The primary endpoint was prostate-cancer specifi c mortality, analysed according to the intention-to-screen principle. The study is ongoing, with men who have not reached the upper age limit invited for PSA testing. This is the fi rst planned report on cumulative prostate-cancer incidence and mortality calculated up to Dec 31, This study is registered as an International Standard Randomised Controlled Trial ISRCTN Findings In each group, 48 men were excluded from the analysis because of death or emigration before therandomisation date, or prevalent prostate cancer. In men randomised to screening, 7578 (76%) of 9952 attended atleast once. During a median follow-up of 14 years, 1138 men in the screening group and 718 in the control group were diagnosed with prostate cancer, resulting in a cumulative prostate-cancer incidence of 12 7% in the screening group and 8 2% in the control group (hazard ratio 1 64; 95% CI ; p<0 0001). The absolute cumulative risk reduction of death from prostate cancer at 14 years was 0 40% (95% CI ), from 0 90% in the control group to 0 50% in the screening group. The rate ratio for death from prostate cancer was 0 56 (95% CI ; p=0 002) in the screening compared with the control group. The rate ratio of death from prostate cancer for attendees compared with the control group was 0 44 (95% CI ; p=0 0002). Overall, 293 (95% CI ) men needed to be invited for screening and 12 to be diagnosed to prevent one prostate cancer death.
31 Mortality results from the Göteborg randomised population-based prostate-cancer screening trial. Hugosson. Lancet Oncology 2011
32 Between 1993 and 2001, of 76,693 men who were randomly assigned to usual care or intervention at 10 US centers, 73,378 (96%) completed a questionnaire that inquired about comorbidity and prostate specific antigen (PSA) testing before random assignment Fig 2. (A) Unadjusted cumulative incidence estimates14 of prostate cancer specific mortality in men with no or minimal comorbidity randomly assigned to usual care or intervention. k sample P.03. (B) Unadjusted cumulative incidence estimates14 of prostate cancer specific mortality in men with at least one significant comorbidity randomly assigned to usual care or intervention. k sample P=.07. Bonferroni adjustment16 for a significant P value was<.05 / 2 or.025 Crawford ED, et al. Comorbidity and mortality results from a randomized prostate cancer screening trial. J Clin Oncol, 2011; 29 (4):
33 Mejora Empeora Bouchardy C, et al. Recent trends in prostate cancer mortality show a continuous decrease in several countries. Int J Cancer 2008; 123:
34 Characteristics and methodological quality of randomised controlled trials of screening for prostate cancer No of participants randomised Study Screened group Control group Age range (years) Screening test Randomisatio n Allocation concealment Descripti on of loss to followup Blinding of outcome assessors Contamina tion (of control group) Intention to screen analysis Approximate median follow up time (years) Quebec PSA+DRE Adequate Not adeuate No Not adequate Not provided Not analysed but data provided 11 Norrkoping DRE initialy; PSA+DRE after 1993 Quasirandomi sation Not adequate Yes Not adequate Not provided Yes 13 (diagnosis) 15(death) ERSPC PSA+DRE Adquate Not adequate Unclear Yes Yes (20%) Yes 9 French ERSPC PSA Unclear Unclear Unclear Unclear Unclear Unclear 4 PLCO PSA+DRE Adequate Adequate Yes Yes Yes (40 52%) Yes 11.5 Gothenbur g PSA Adequate Not adequate Yes Yes Low, details not provided Yes 14 Djulbegovic M, et al. Screening for prostate cancer: systematic review and metaanalysis of randomised controlled trials. BMJ 2010; 341: c4543
35 Context Examining the tradeoffs between potential benefits and harms of prostate cancer screening is a hot topic. Contribution This updated systematic review for the U.S. Preventive Services Task Force found the following: screening based on prostate specific antigen led to detection of more cases of prostate cancer, small to no reduction in prostate cancer specific mortality after about 10 years, and several potential harms related to falsepositive test results and subsequent evaluations and therapies Caution Evidence regarding the mortality associated benefits of screening conflicted. Implication The clinical benefits of screening for prostate cancer remain uncertain. Consequences include evaluations and treatments that have associated complications and that may be unnecessary. Chou R, et al. Screening for Prostate Cancer: A Review of the Evidence for the U.S. Preventive Services Task Force. Ann Intern Med 2011; 155:
36 Summary of findings in trials on screening for prostate cancer Illustrative comparative risks (95% CI) Outcomes Event rate (per 1000) without screening Event rate (per 1000) with screening Relative risk (95% CI) No of participants Quality of evidence (GRADE) All cause mortality (inverse variance) Death from prostate cancer (inverse variance) Prostate cancer diagnosis (194 to 202) 0.99 (0.97 to 1.01) Moderate Moderate Low Effects of screening on stage: Stage I Stage II Stage III IV (13 to 34) 32 (23 to 45) 5 (4 to 5) 1.95 (1.22 to 3.13) 1.39 (0.99 to 1.95) 0.94 (0.85 to 1.04) Low Very low Moderate Djulbegovic M, et al. Screening for prostate cancer: systematic review and metaanalysis of randomised controlled trials. BMJ 2010; 341: c4543
37 Wilt TJ, et al. Radical prostatectomy versus observation for localized prostate cancer. N Engl J Med. 2012; 367 (3): PIVOT BACKGROUND The effectiveness of surgery versus observation for men with localized prostate cancer detected by means of prostate specific antigen (PSA) testing is not known. METHODS From November 1994 through January 2002, we randomly assigned 731 men with localized prostate cancer (mean age, 67 years; median PSA value, 7.8 ng per milliliter) to radical prostatectomy or observation and followed them through January The primary outcome was all cause mortality; the secondary outcome was prostatecancer mortality. Controversias Tratamiento el screening del cáncer del cáncer próstata de localizado próstata: y A localmente quién y cuándo avanzado debo hacer un PSA?
38 RESULTS Radical prostatectomy was associated with reduced all cause mortality among men with a PSA value greater than 10 ng per milliliter (P = 0.04 for interaction) and possibly among those with intermediate risk or high risk tumors (P = 0.07 for interaction). Adverse events within 30 days after surgery occurred in 21.4% of men, including one death. CONCLUSIONS Among men with localized prostate cancer detected during the early era of PSA testing, radical prostatectomy did not significantly reduce all cause or prostate cancer mortality, as compared with observation, through at least 12 years of follow up. Absolute differences were less than 3 percentage points. (Funded by the Department of Veterans Affairs Cooperative Studies Program and others; PIVOT ClinicalTrials.gov number, NCT ) Wilt TJ, et al. Radical prostatectomy versus observation for localized prostate cancer. N Engl J Med. 2012; 367 (3): Controversias Tratamiento el screening del cáncer del cáncer próstata de localizado próstata: y A localmente quién y cuándo avanzado debo hacer un PSA?
39 Figure 1. Study enrollment and treatment Wilt TJ, et al. Radical prostatectomy versus observation for localized prostate cancer. N Engl J Med. 2012; 367 (3): Controversias Tratamiento el screening del cáncer del cáncer próstata de localizado próstata: y A localmente quién y cuándo avanzado debo hacer un PSA?
40 Figure S2c-e. All-casue mortality by risk category: c: low risk; d: intermediate risk; e: hig risk Supplement to: Wilt TJ, Brawer MK, Jones KM, et al. Radical prostatectomy versus observation for localized prostate cancer. N Engl J Med 2012;367: OI: /NEJMoa Controversias Tratamiento el screening del cáncer del cáncer próstata de localizado próstata: y A localmente quién y cuándo avanzado debo hacer un PSA?
41 Flowchart of the comparison arm for ProtecT trial Lane JA, et al. Latest results from the UK trials evaluating prostate cancer screening and treatment: The CAP and ProtecT studies. Eur J Cancer 2010; 46:
42 . Design characteristics of the CAP trial and ERSPC TRIAL CHARACTERISTIC CAP TRIAL ERSPC Randomisation unit Location Eligibility criteria Recruitment period Recruitment total Intervention group: PSA threshold (total PSA) DRE with PSA testing Biopsy protocol Rescreening interval Treatment Control group Outcome ascertainment Primary outcome Follow up Quality of life Resource use Primary care centre United Kingdom Menaged50 60 years >550 (ca. 450,000 individuals) 3.0 ng/ml No 10 cores Not in protocol Localised disease: randomisation or choice of active monitoring, radiotherapy or surgery Advanced disease: local policy Usual clinical care Independent blinded adjudication committee Prostate cancer specific mortality Average 10 years (up to 2016) Across intervention group Across both groups Individual 7 European countries Menaged55 69 years ,243 individuals 3.0 ng/ml ( 4.0) Yes 6cores 4 years (2 7) Local policy Usual clinical care Independent blinded adjudication committee Prostate cancer specific mortality Median 9 years (up to 2007) Several centres Several centres Lane JA, et al. Latest results from the UK trials evaluating prostate cancer screening and treatment: The CAP and ProtecT studies. Eur J Cancer 2010; 46:
43 ESPECTATIVA DE VIDA EDAD ESPERANZA DE VIDA 37,11 32,08 27,70 23,48 19,50 15,71 12,21 9,16 6,65 4,75 3,36 2,61 Fuente: www. Ine.es acceso enero 2011
44 CLASIFICACIÓN Y VALORACIÓN DE RIESGO SEGÚN LAS COMORBILIDADES EN PACIENTES CANDIDATOS A CIRUGÍA. VALORACIÓN DEL RIESGO QUIRÚRGICO. Índice de Charlson Se considera: Ausencia de comorbilidad: 0-1 puntos Comorbilidad baja: 2 puntos Comorbilidad alta: > 3 puntos. Predicción de mortalidad/año a 3 años: Índice de 0: 12% Índice 1-2: 26% Índice 3-4: 52% Índice > 5: 85%
45 No se puede mostrar la imagen en este momento. DEFINIR POBLACIONES DE TUMORES Introducción Dr necesito hacer una biopsia?
46
47 MODELOS Análisis de los resultados aplicando los nomogramas Autor Referencia Año Nº ptes PSA rango (ng/ml) Bx/Nº cilindros 1ª bx/otras ABC Modelo Thompson J Natl Cancer Inst. 2006; 98, ,3-287? Múltiples 0,72 Regresión logística Nam J Clin Oncol. 2007; 25: , ? 0,74 cáncer 0,77 alto riesgo Regresión logística Kawakami Eur Urol. 2008; 54: <20 Media 22 0,75 cáncer 0,80 alto riesgo ANN Chun Eur Urol. 2007; 52: ? 0,8 Estudio J Urol , º 0,76 cáncer 0,82 alto riesgo 0,78 cilindros (+) Regresión logística Regresión logística
48 2147 pacientes PSA>3 Biopsia: sextante ABC: 0,77 Roobol MJ, et al. A Risk Based Strategy Improves Prostate Specific Antigen Driven Detection of Prostate Cancer. Eur Urol 2010, 79 85
49
50 Age-Adjusted Incidence Rates of Prostate Cancer Over Time and by Prescreen and Postcreen Snapshot Esseman L, et al. Rethinking screening for breast cancer and prostate cancer. JAMA. 2009; 302 (15):
51 After 20 years of screening for breast and prostate cancer, several observations can be made. First, the incidence of these cancers increased after the introduction of screening but has never returned to prescreening levels. Second, the increase in the relative fraction of early stage cancers has increased. Third, the incidence of regional cancers has not decreased at a commensurate rate. One possible explanation is that screening may be increasing the burden of low risk cancers without significantly reducing the burden of more aggressively growing cancers and therefore not resulting in the anticipated reduction in cancer mortality. To reduce morbidity and mortality from prostate cancer and breast cancer, new approaches for screening, early detection, and prevention for both diseases should be considered. Esseman L, et al. Rethinking screening for breast cancer and prostate cancer. JAMA. 2009; 302 (15):
52 Screen Detection Capability Based on Tumor Biology and Growth Rates Esseman L, et al. Rethinking screening for breast cancer and prostate cancer. JAMA. 2009; 302 (15):
53 Zeliadt SB, et al. Influence of Publication of US and European Prostate Cancer Screening Trials on PSA Testing Practices. J Natl Cancer Inst. 2011; 103(6):
54 Registro Nacional de cáncer de Próstata 2010 en España J M.Cozar, Miñana, G- Veiga. A Rodriguez et al. BJU int 2012 and Actas Españolas de Urologia 2012
55 Registro Nacional de cáncer de Próstata 2010 en España J M.Cozar, Miñana, G- Veiga. A Rodriguez et al. BJU int 2012 and Actas Españolas de Urologia 2012 Enfermedad Cardio-Vascular: 1968(48.15%)
56 Percentage of Voters in a Region in Favor of Prostate-Specific Antigen (PSA) Testing, According to Geographic Region. Colbert JA, et al. Prostate Cancer Screening Polling Results. N Engl J Med. 2012
57 Provisional Clinical Opinion On the basis of identified evidence and the expert opinion of the panel: In men with a life expectancy<10 years,* it is recommended that general screening for prostate cancer with total PSA be discouraged, because harms seem to outweigh potential benefits. Type and strength of recommendation. Evidence based: strong. Strength of evidence. Moderate:basedonfive randomizedclinicaltrials (RCTs) with intermediate to high risk of bias, moderate follow up, and limited data on subgroup populations. In men with a life expectancy>10 years,* it is recommended that physicians discuss with their patients whether PSA testing for prostate cancer screening is appropriate for them. PSA testing may save lives but is associated with harms, including complications, from unnecessary biopsy, surgery, or radiation treatment. Type and strength of recommendation. Evidence based: strong. Strength of evidence. For benefit, moderate; for harm, strong: based on five RCTs (and several cohort studies) with intermediate to high risk of bias, moderate follow up, indirect data, inconsistent results, and limited data on subgroup populations. It is recommended that information written in lay language be available to clinicians and their patients to facilitate the discussion of the benefits and harms associated with PSA testing before the routine ordering of a PSA test. Type and strength of recommendation. Informal consensus: strong. Strength of evidence. Indeterminate: evidence was not systematically reviewed to inform this recommendation; however, randomized trials are available on the topic. *Calculation of life expectancy is based on a variety of individual factors and circumstances. A number of life expectancy calculators (eg, are available in the public domain; however, ASCO does not endorse any one calculator over another. Basch E, et al. Screening for Prostate Cancer With Prostate-Specific Antigen Testing: American Society of Clinical Oncology Provisional Clinical Opinion. J Clin Oncol. 2012; 30 (24):
58 EUA (1) AUA (2) NCCN (3) Sociedad Americana Cancer (4) US TASK FORCE (5) Año Recomendación de Cribado sistemático No No No No Detección Precoz Edad Antecedentes Familiares Tacto rectal PSA (indicación biopsia) 2 3 2,5 4 2,5 4 2,5 4 Factores Riesgo PSA % PSAc/t <25 PSAv>0,75 ng/ml PSAv>0,75ng/ml Factores de Agresividad No No No No Técnica de biopsia Ecodirigida Ecodirigida Ecodirigida Ecodirigida Nº de punciones Edad límite >75 Expectativa <10 años Comorbilidades valoración Decisión de cribado y Biopsia >75* Expectativa <10 años Informada y compartida Informada y compartida Informada y compartida Informada y compartida
59 Case 1 PSA Biopsy: Gleason 7 (3+4), 9c+/10 90% + c. 47 year old man 1st PSA: 1.7ng/ml Rectal E.: Slightly fibrous Prostate volume: 22 cc Weight: 72kg Height: 1.72m. No Comorbidities Family history of PC
60 CONCLUSIONES El PSA reduce muertes por cáncer de próstata. El nivel inicial de PSA debe ser inferior a 4, 2,5-3 en pacientes con expectativa de vida mayor de 10 años. Las características del paciente son fundamentales para indicar test. La edad en la que recomendar no hacer determinación no está definida pero en varones mayores de 75 no parece ser beneficioso. Los modelos nos pueden ayudar a tomar la decisión con el paciente Nuevos marcadores están en marcha, pero no sustituyen al PSA en la actualidad. Importante informar y consensuar con el paciente riesgos-beneficios, Screening.no, despistaje sí...
61 Gracias
Cáncer de próstata. Paradigmas que cambian en 2013, por Mauricio Lema Medina
 Cáncer de próstata Paradigmas que cambian en 2013, por Mauricio Lema Medina Nuevos paradigmas en cáncer de próstata (CaPr) Temario 1 2 3 4 5 USPSTF recomienda NO practicar tamizaje con PSA Observación,
Cáncer de próstata Paradigmas que cambian en 2013, por Mauricio Lema Medina Nuevos paradigmas en cáncer de próstata (CaPr) Temario 1 2 3 4 5 USPSTF recomienda NO practicar tamizaje con PSA Observación,
Prevención Cuaternaria. Cribado de cáncer de mama Uruguay
 Prevención Cuaternaria Cribado de cáncer de mama Uruguay 1 2 3 OPORTUNIDAD O AMENAZA? 4 Ley carne de salud Control periódico toda/os los trabajadores. En mujeres se exige realización de: PAP Mamografía
Prevención Cuaternaria Cribado de cáncer de mama Uruguay 1 2 3 OPORTUNIDAD O AMENAZA? 4 Ley carne de salud Control periódico toda/os los trabajadores. En mujeres se exige realización de: PAP Mamografía
Influencia de las dislipemias y de su tratamiento sobre el metabolismo de la glucosa, la función renal y miocárdica
 Influencia de las dislipemias y de su tratamiento sobre el metabolismo de la glucosa, la función renal y miocárdica Dr. Carlos Guijarro Unidad de Medicina Interna Universidad Rey Juan Carlos Dislipemias,
Influencia de las dislipemias y de su tratamiento sobre el metabolismo de la glucosa, la función renal y miocárdica Dr. Carlos Guijarro Unidad de Medicina Interna Universidad Rey Juan Carlos Dislipemias,
El gen PCA3, un nuevo biomarcador diagnóstico y predictivo del grado de Gleason y el volumen tumoral en el carcinoma de próstata
 El gen PCA3, un nuevo biomarcador diagnóstico y predictivo del grado de Gleason y el volumen tumoral en el carcinoma de próstata Natalia Rodón et al. BIOPAT Biopatología Molecular S.L. Grup Assistència
El gen PCA3, un nuevo biomarcador diagnóstico y predictivo del grado de Gleason y el volumen tumoral en el carcinoma de próstata Natalia Rodón et al. BIOPAT Biopatología Molecular S.L. Grup Assistència
PSA Y TACTO RECTAL EN EL CRIBADO DE CÁNCER DE PRÓSTATA
 PSA Y TACTO RECTAL EN EL CRIBADO DE CÁNCER DE PRÓSTATA Tutor: Josep Casajuana Brunet, Médico rural en Tarragona Ponente: Verónica Valle Gallardo, Residente de MFyC en Cádiz IDENTIFICAR EL PROBLEMA Se ha
PSA Y TACTO RECTAL EN EL CRIBADO DE CÁNCER DE PRÓSTATA Tutor: Josep Casajuana Brunet, Médico rural en Tarragona Ponente: Verónica Valle Gallardo, Residente de MFyC en Cádiz IDENTIFICAR EL PROBLEMA Se ha
La EPOC como factor de riesgo para Cáncer de Pulmón. Dr. Juan Pablo de Torres Tajes Servicio de Neumología Clínica Universidad de Navarra
 La EPOC como factor de riesgo para Cáncer de Pulmón Dr. Juan Pablo de Torres Tajes Servicio de Neumología Clínica Universidad de Navarra Resumen Una asociación letal Nuestros datos Posibles mecanismos
La EPOC como factor de riesgo para Cáncer de Pulmón Dr. Juan Pablo de Torres Tajes Servicio de Neumología Clínica Universidad de Navarra Resumen Una asociación letal Nuestros datos Posibles mecanismos
Actualización Médica Periódica Número 93 www.ampmd.com Febrero 2009
 Actualización Médica Periódica Número 93 www.ampmd.com Febrero 2009 TÓPICOS EN ATENCIÓN PRIMARIA Dr. José Agustín Arguedas Quesada ANTÍGENO PROSTÁTICO ESPECÍFICO Introducción El carcinoma de la próstata
Actualización Médica Periódica Número 93 www.ampmd.com Febrero 2009 TÓPICOS EN ATENCIÓN PRIMARIA Dr. José Agustín Arguedas Quesada ANTÍGENO PROSTÁTICO ESPECÍFICO Introducción El carcinoma de la próstata
Traducido por: Dr. Pedro A. Vargas Torres Obstetra Ginecólogo
 The American College of Obstetricians and Gynecologists WOMEN S HEALTH CARE PHYSICIANS Traducido por: Dr. Pedro A. Vargas Torres Obstetra Ginecólogo NOTA: Esta es una traducción libre realizada con la
The American College of Obstetricians and Gynecologists WOMEN S HEALTH CARE PHYSICIANS Traducido por: Dr. Pedro A. Vargas Torres Obstetra Ginecólogo NOTA: Esta es una traducción libre realizada con la
"Average years of healthy life (without disabilities). Argentina 2002-2003.
 Universidad Nacional de Córdoba "Average years of healthy life (without disabilities). Argentina 2002-2003. Alvarez, M. - Carrizo, E. - Peláez, E. - González, L. CONICET - National University of Córdoba,
Universidad Nacional de Córdoba "Average years of healthy life (without disabilities). Argentina 2002-2003. Alvarez, M. - Carrizo, E. - Peláez, E. - González, L. CONICET - National University of Córdoba,
Cirugía radical de próstata?
 ESP DECIPHER HABILITA A UN TRATAMIENTO PERSONALIZADO Y PROCESABLE DESPUÉS DE LA CIRUGÍA Cirugía radical de próstata? El Clasificador de Cáncer de Próstata Decipher puede ayudarlos a usted y a su médico
ESP DECIPHER HABILITA A UN TRATAMIENTO PERSONALIZADO Y PROCESABLE DESPUÉS DE LA CIRUGÍA Cirugía radical de próstata? El Clasificador de Cáncer de Próstata Decipher puede ayudarlos a usted y a su médico
Hígado bioartificial Revisión sistemática
 CONSEJERÍA DE IGUALDAD, SALUD Y POLÍTICAS SOCIALES AGENCIA DE EVALUACIÓN DE TECNOLOGÍAS SANITARIAS DE ANDALUCÍA (AETSA) Hígado bioartificial Revisión sistemática Informe de evaluación de tecnologías emergentes
CONSEJERÍA DE IGUALDAD, SALUD Y POLÍTICAS SOCIALES AGENCIA DE EVALUACIÓN DE TECNOLOGÍAS SANITARIAS DE ANDALUCÍA (AETSA) Hígado bioartificial Revisión sistemática Informe de evaluación de tecnologías emergentes
Alejandra Zavala Molina * Carlos Meza Galo **
 156 Utilidad clínica de la relación antígeno prostático específico libre (psal)/ antígeno prostático específico total (psat) en el diagnóstico de cáncer de próstata. Clinical utility of free prostate specific
156 Utilidad clínica de la relación antígeno prostático específico libre (psal)/ antígeno prostático específico total (psat) en el diagnóstico de cáncer de próstata. Clinical utility of free prostate specific
Evidencias del screening de cáncer de próstata con PSA
 EDITORIAL EDITORIAL Rev. Arg. de Urol. Vol. 77 (3) 2012 (152-156) ISSN 0327-3326 Evidencias del screening de cáncer de próstata con PSA Evidence in prostate cáncer screening with PSA Juan Ignacio Monzó
EDITORIAL EDITORIAL Rev. Arg. de Urol. Vol. 77 (3) 2012 (152-156) ISSN 0327-3326 Evidencias del screening de cáncer de próstata con PSA Evidence in prostate cáncer screening with PSA Juan Ignacio Monzó
BIOPSIA DE PRÓSTATA. Dr. R. Ferrero Doria. Urología Hospital Denia
 BIOPSIA DE PRÓSTATA Dr. R. Ferrero Doria Urología Hospital Denia INTRODUCCIÓN Métodos que nos dirigen hacia la B.P. Ecografía transrectal Tacto rectal PSA ANTIGENO PROSTÁTICO ESPECÍFICO PSA es el verdaderamente
BIOPSIA DE PRÓSTATA Dr. R. Ferrero Doria Urología Hospital Denia INTRODUCCIÓN Métodos que nos dirigen hacia la B.P. Ecografía transrectal Tacto rectal PSA ANTIGENO PROSTÁTICO ESPECÍFICO PSA es el verdaderamente
Recomendaciones de cribado en cáncer de próstata en el año 2010 / Prostate cancer screening guidelines in 2010 Dr. Marcos Luján, Servicio de
 Recomendaciones de cribado en cáncer de próstata en el año 2010 / Prostate cancer screening guidelines in 2010 Dr. Marcos Luján, Servicio de Urología, Hospital Infanta Cristina, Madrid Introducción. El
Recomendaciones de cribado en cáncer de próstata en el año 2010 / Prostate cancer screening guidelines in 2010 Dr. Marcos Luján, Servicio de Urología, Hospital Infanta Cristina, Madrid Introducción. El
Cáncer de mama epidemiologia descriptiva, factores de riesgo y cribado
 Cáncer de mama epidemiologia descriptiva, factores de riesgo y cribado Josep A Espinàs Plan director de oncología de Cataluña EPIDEMIOLOGIA DESCRIPTIVA ECO-EUCAN (Update minor) (21 Jan. 2015) Texto 1 Texto
Cáncer de mama epidemiologia descriptiva, factores de riesgo y cribado Josep A Espinàs Plan director de oncología de Cataluña EPIDEMIOLOGIA DESCRIPTIVA ECO-EUCAN (Update minor) (21 Jan. 2015) Texto 1 Texto
Patters of evolution of the Mexican clearing house system (1940-1980) Demography or Levels of Economic Activity? Gustavo A.
 Patters of evolution of the Mexican clearing house system (1940-1980) Demography or Levels of Economic Activity? Gustavo A. Del Angel 2 Some facts for the period 1940-1980: Between the 1940s and the late
Patters of evolution of the Mexican clearing house system (1940-1980) Demography or Levels of Economic Activity? Gustavo A. Del Angel 2 Some facts for the period 1940-1980: Between the 1940s and the late
Terapia de campo de tumores en el tratamiento del glioblastoma
 Terapia de campo de tumores en el tratamiento del glioblastoma Revisión sistemática Informe de síntesis de tecnología emergente Tumor treating fields therapy (TTF) for glioblastoma. A Systematic Review
Terapia de campo de tumores en el tratamiento del glioblastoma Revisión sistemática Informe de síntesis de tecnología emergente Tumor treating fields therapy (TTF) for glioblastoma. A Systematic Review
Tratamiento prolongado antiagregante en subgrupos de pacientes. José F. Díaz, MD, FESC Complejo Hospitalario de Huelva
 Tratamiento prolongado antiagregante en subgrupos de pacientes José F. Díaz, MD, FESC Complejo Hospitalario de Huelva Endpoint (%) Primary Endpoint CV Death,MI,Stroke 15 10 CV Death / MI / Stroke Clopidogrel
Tratamiento prolongado antiagregante en subgrupos de pacientes José F. Díaz, MD, FESC Complejo Hospitalario de Huelva Endpoint (%) Primary Endpoint CV Death,MI,Stroke 15 10 CV Death / MI / Stroke Clopidogrel
OPTIMIZACIÓN TRATAMIENTO ANTI-EGFR. Ruth Vera Oncología Médica
 OPTIMIZACIÓN TRATAMIENTO ANTI-EGFR Ruth Vera Oncología Médica OPTIMIZACIÓN TRATAMIENTO anti-egfr OPTIMIZAR quiere decir: Buscar los mejores resultados Planificar una actividad para obtener los mejores
OPTIMIZACIÓN TRATAMIENTO ANTI-EGFR Ruth Vera Oncología Médica OPTIMIZACIÓN TRATAMIENTO anti-egfr OPTIMIZAR quiere decir: Buscar los mejores resultados Planificar una actividad para obtener los mejores
Incertidumbres en el manejo del PSA
 Incertidumbres en el manejo del PSA Kiko Brotons Muntó CS. Vila-real II (Carinyena) GdT Nefro-urología semfyc 1970 (MCMLXX) Richard J. Ablin y el PSA (2010) I never dreamed that my discovery four decades
Incertidumbres en el manejo del PSA Kiko Brotons Muntó CS. Vila-real II (Carinyena) GdT Nefro-urología semfyc 1970 (MCMLXX) Richard J. Ablin y el PSA (2010) I never dreamed that my discovery four decades
Improving Rates of Colorectal Cancer Screening Among Never Screened Individuals
 Improving Rates of Colorectal Cancer Screening Among Never Screened Individuals Northwestern University, Feinberg School of Medicine Contents Patient Letter Included with Mailed FIT... 3 Automated Phone
Improving Rates of Colorectal Cancer Screening Among Never Screened Individuals Northwestern University, Feinberg School of Medicine Contents Patient Letter Included with Mailed FIT... 3 Automated Phone
CÁNCER PRÓSTATA LOCALIZADO SBRT vs Braquiterapia (HDR) HIPOFRACCIONAMIENTO EXTREMO
 CÁNCER PRÓSTATA LOCALIZADO SBRT vs Braquiterapia (HDR) HIPOFRACCIONAMIENTO EXTREMO Qué diferencia ves? SBRT vs HDR DEFINICIÓN: SBRT VS HDR HIPOFRACCIONAMIENTO EXTREMO PLATFORM SBRT PROCEDIMIENTO SBRT CARACTERÍSTICAS
CÁNCER PRÓSTATA LOCALIZADO SBRT vs Braquiterapia (HDR) HIPOFRACCIONAMIENTO EXTREMO Qué diferencia ves? SBRT vs HDR DEFINICIÓN: SBRT VS HDR HIPOFRACCIONAMIENTO EXTREMO PLATFORM SBRT PROCEDIMIENTO SBRT CARACTERÍSTICAS
MAGNITUD DEL PROBLEMA
 CÁNCER DE PRÓSTATA JUSTIFICACIÓN El cáncer de próstata (CP) es un problema de salud que suscita un interés creciente, tanto entre la población general como entre los profesionales sanitarios. Algunos de
CÁNCER DE PRÓSTATA JUSTIFICACIÓN El cáncer de próstata (CP) es un problema de salud que suscita un interés creciente, tanto entre la población general como entre los profesionales sanitarios. Algunos de
Detección Precoz de Cáncer de
 [REV. MED. CLIN. CONDES - 2013; 24(4) 654-659] Detección Precoz de Cáncer de Próstata Early detection of prostate cancer Dr. Christian Ramos G. (1), Dr. Juan Fullá O. (1) 1. Departamento de Urología. Clínica
[REV. MED. CLIN. CONDES - 2013; 24(4) 654-659] Detección Precoz de Cáncer de Próstata Early detection of prostate cancer Dr. Christian Ramos G. (1), Dr. Juan Fullá O. (1) 1. Departamento de Urología. Clínica
Thank you. US English US Spanish. Australia-English Canada-English Ireland-English New Zealand-English Taiwan-English United Kingdom-English
 Dear Healthcare Provider, Included in this PDF are recruitment brochures in several languages to be used in MM Bone study (Protocol No.: 20090482). Kindly note these brochures have been updated according
Dear Healthcare Provider, Included in this PDF are recruitment brochures in several languages to be used in MM Bone study (Protocol No.: 20090482). Kindly note these brochures have been updated according
Pregunta 1 Suponga que una muestra de 35 observaciones es obtenida de una población con media y varianza. Entonces la se calcula como.
 Universidad de Costa Rica Programa de Posgrado en Computación e Informática Doctorado en Computación e Informática Curso Estadística 18 de febrero 2013 Nombre: Segundo examen corto de Probabilidad Pregunta
Universidad de Costa Rica Programa de Posgrado en Computación e Informática Doctorado en Computación e Informática Curso Estadística 18 de febrero 2013 Nombre: Segundo examen corto de Probabilidad Pregunta
José María Hernández
 José María Hernández 1.Introduction 2.PARTNER Trials 3.COREVALVE Trials 4.Current Guidelines 5.Intermediate Risk 6.Low Risk 7.Conclusions < 2008 21 34 Rigorous Clinical Research PubMed annual number TAVI
José María Hernández 1.Introduction 2.PARTNER Trials 3.COREVALVE Trials 4.Current Guidelines 5.Intermediate Risk 6.Low Risk 7.Conclusions < 2008 21 34 Rigorous Clinical Research PubMed annual number TAVI
Prostate Cancer. The need to urinate more often, especially at night. A hard time starting to urinate or holding back urine
 Prostate Cancer The prostate is a gland in men that makes a liquid that forms part of semen. Its size is about 2 centimeters by 2 centimeters and is located in the pelvic area. The prostate uses a male
Prostate Cancer The prostate is a gland in men that makes a liquid that forms part of semen. Its size is about 2 centimeters by 2 centimeters and is located in the pelvic area. The prostate uses a male
EL CRIBADO DEL CANCER DE PRÓSTATA. Situación del cáncer de próstata en España
 EL CRIBADO DEL CANCER DE PRÓSTATA. Situación del cáncer de próstata en España El cáncer de próstata es el tumor más frecuente en los hombres españoles, con más de 25000 nuevos diagnósticos anuales 1. Cuatro
EL CRIBADO DEL CANCER DE PRÓSTATA. Situación del cáncer de próstata en España El cáncer de próstata es el tumor más frecuente en los hombres españoles, con más de 25000 nuevos diagnósticos anuales 1. Cuatro
PROBLEMAS PARA LA CLASE DEL 20 DE FEBRERO DEL 2008
 PROBLEMAS PARA LA CLASE DEL 20 DE FEBRERO DEL 2008 Problema 1 Marketing estimates that a new instrument for the analysis of soil samples will be very successful, moderately successful, or unsuccessful,
PROBLEMAS PARA LA CLASE DEL 20 DE FEBRERO DEL 2008 Problema 1 Marketing estimates that a new instrument for the analysis of soil samples will be very successful, moderately successful, or unsuccessful,
Caso clínico: Mujer diabética de 82 años
 Caso clínico: Mujer diabética de 82 años José Manuel Millaruelo Trillo Centro de Salud Torrero La Paz. Zaragoza Reunión GEDAPS Barcelona 6 de Noviembre 2009 Posibles enfoques Encorsetarla en los objetivos
Caso clínico: Mujer diabética de 82 años José Manuel Millaruelo Trillo Centro de Salud Torrero La Paz. Zaragoza Reunión GEDAPS Barcelona 6 de Noviembre 2009 Posibles enfoques Encorsetarla en los objetivos
Where are Chilean companies hiring?
 Where are Chilean companies hiring? Abstract: Taking advantage that unemployment figures are low, it is common to wonder what sectors of the economy are hiring more. So if you are looking for a job in
Where are Chilean companies hiring? Abstract: Taking advantage that unemployment figures are low, it is common to wonder what sectors of the economy are hiring more. So if you are looking for a job in
La mortalidad inmediata total fue de 3,3%. La frecuencia de mortalidad para los grupos fue de 6,5% (n= 6) vs. 1,9% (n= 4) respectivamente.
 33 Bogotá, Colombia. Existen controversias acerca de las posibles ventajas del abordaje transperitoneal vs. extraperitoneal en la cirugía de aneurisma de aorta abdominal; con este último, algunos estudios
33 Bogotá, Colombia. Existen controversias acerca de las posibles ventajas del abordaje transperitoneal vs. extraperitoneal en la cirugía de aneurisma de aorta abdominal; con este último, algunos estudios
Brain Tumors. Glioma Tumors
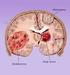 Brain Tumors A tumor that starts and grows in the brain is called a primary brain tumor. Primary brain tumors get their name from the type of cell or the part of the brain where they start growing. A secondary
Brain Tumors A tumor that starts and grows in the brain is called a primary brain tumor. Primary brain tumors get their name from the type of cell or the part of the brain where they start growing. A secondary
4Kscore la prueba de sangre que identifica el riesgo de cáncer de próstata agresivo
 4Kscore la prueba de sangre que identifica el riesgo de cáncer de próstata agresivo Detección temprana del cáncer de próstata agresivo Desafíos El PSA total tiene un alta tasa de falsos positivos Más de
4Kscore la prueba de sangre que identifica el riesgo de cáncer de próstata agresivo Detección temprana del cáncer de próstata agresivo Desafíos El PSA total tiene un alta tasa de falsos positivos Más de
VIGILANCIA ACTIVA EN CANCER DE PROSTATA
 VIGILANCIA ACTIVA EN CANCER DE PROSTATA Dr. Levin Martínez Prof. Agregado Titular de Urología Facultad de Medicina-Uruguay XX Workshop Urologia Oncológica 17 18 Abril 2015 Carvoeiro. Algarve Definir VA
VIGILANCIA ACTIVA EN CANCER DE PROSTATA Dr. Levin Martínez Prof. Agregado Titular de Urología Facultad de Medicina-Uruguay XX Workshop Urologia Oncológica 17 18 Abril 2015 Carvoeiro. Algarve Definir VA
Análisis de la coestimulación vía CD28 en células linfoides de pacientes infectados. con el virus de la Hepatitis C RESUMEN
 Análisis de la coestimulación vía CD28 en células linfoides de pacientes infectados con el virus de la Hepatitis C RESUMEN La infección por el virus de la hepatitis C (VHC) afecta a más de 170 millones
Análisis de la coestimulación vía CD28 en células linfoides de pacientes infectados con el virus de la Hepatitis C RESUMEN La infección por el virus de la hepatitis C (VHC) afecta a más de 170 millones
UTILIDAD REAL DE NOMOGRAMAS EN UROLOGÍA. Mario Paúl Sánchez Pérez MIR 2º año Urología Centro Hospitalario Universitario de Badajoz
 UTILIDAD REAL DE NOMOGRAMAS EN UROLOGÍA Mario Paúl Sánchez Pérez MIR 2º año Urología Centro Hospitalario Universitario de Badajoz Nomograma Representación gráfica que permite realizar con rapidez cálculos
UTILIDAD REAL DE NOMOGRAMAS EN UROLOGÍA Mario Paúl Sánchez Pérez MIR 2º año Urología Centro Hospitalario Universitario de Badajoz Nomograma Representación gráfica que permite realizar con rapidez cálculos
Tratamiento de la hiperplasia benigna de próstata mediante enucleación con láser holmio
 Tratamiento de la hiperplasia benigna de próstata mediante enucleación con láser holmio CT2009/01 RESUMEN Introducción La hiperplasia benigna de próstata (HBP) se caracteriza por aumento del tamaño glandular,
Tratamiento de la hiperplasia benigna de próstata mediante enucleación con láser holmio CT2009/01 RESUMEN Introducción La hiperplasia benigna de próstata (HBP) se caracteriza por aumento del tamaño glandular,
Identificación rápida de Staphylococcus aureus meticilina resistente Cribado de portadores
 Identificación rápida de Staphylococcus aureus meticilina resistente Cribado de portadores Informe de síntesis de tecnologías emergentes Rapid tests for methicillin resistant Staphylococcus aureus. Carrier
Identificación rápida de Staphylococcus aureus meticilina resistente Cribado de portadores Informe de síntesis de tecnologías emergentes Rapid tests for methicillin resistant Staphylococcus aureus. Carrier
New Horizonts for the management of Dyslipidemia. LDL-c Less than 70 mg/dl
 New Horizonts for the management of Dyslipidemia. LDL-c Less than 70 mg/dl Luis Masana Vascular Medicine and Metabolic Unit Sant Joan University Hospital Rovira & Virgili University. IISPV CIBERDEM Reus
New Horizonts for the management of Dyslipidemia. LDL-c Less than 70 mg/dl Luis Masana Vascular Medicine and Metabolic Unit Sant Joan University Hospital Rovira & Virgili University. IISPV CIBERDEM Reus
QUE ES CANCER DE MAMA PDF
 QUE ES CANCER DE MAMA PDF ==> Download: QUE ES CANCER DE MAMA PDF QUE ES CANCER DE MAMA PDF - Are you searching for Que Es Cancer De Mama Books? Now, you will be happy that at this time Que Es Cancer De
QUE ES CANCER DE MAMA PDF ==> Download: QUE ES CANCER DE MAMA PDF QUE ES CANCER DE MAMA PDF - Are you searching for Que Es Cancer De Mama Books? Now, you will be happy that at this time Que Es Cancer De
Certificación en España según normas UNE-EN-ISO 9000 y 14000
 Certificación en España según normas UNE-EN-ISO 9000 y 14000 Sexto informe de Forum Calidad Es este el sexto informe que Forum Calidad presenta sobre el número y distribución de las organizaciones españolas
Certificación en España según normas UNE-EN-ISO 9000 y 14000 Sexto informe de Forum Calidad Es este el sexto informe que Forum Calidad presenta sobre el número y distribución de las organizaciones españolas
SPANISH ORAL LANGUAGE ASSESSMENT. Jill Jegerski Department of Modern Languages April 8, 2011
 SPANISH ORAL LANGUAGE ASSESSMENT Jill Jegerski Department of Modern Languages April 8, 2011 INTRODUCTION Basic Spanish program at CSI Three-course Gen. Ed. sequence: SPN 113, 114, 213 Approximately 800
SPANISH ORAL LANGUAGE ASSESSMENT Jill Jegerski Department of Modern Languages April 8, 2011 INTRODUCTION Basic Spanish program at CSI Three-course Gen. Ed. sequence: SPN 113, 114, 213 Approximately 800
PERSISTENT ORGANIC COMPOUNDS (POC S) ARE VERY TOXIC AND THEY PROVOKE DISEASES LIKE: -- Genetics damage from the birth.
 1 COMPARATIVE STUDY OF THE BIOACUMULATION OF POC S AND GENETIC DAMAGE IN LIVER OF Goodea atripinnis FISH And THE PELECAN Pelicanus erythrorhyncus OF THE LAKE OF CHAPALA AND OF THE LAGOON OF SAYULA According
1 COMPARATIVE STUDY OF THE BIOACUMULATION OF POC S AND GENETIC DAMAGE IN LIVER OF Goodea atripinnis FISH And THE PELECAN Pelicanus erythrorhyncus OF THE LAKE OF CHAPALA AND OF THE LAGOON OF SAYULA According
2015 16 Student Eligibility Verification Advanced Placement/International Baccalaureate Test Fee Program
 2015 16 Student Eligibility Verification Advanced Placement (AP) and/or International Baccalaureate (IB) Exams AP Exam IB Exam AP and IB Exams I. Student Information Last Name First Name MI Grade High
2015 16 Student Eligibility Verification Advanced Placement (AP) and/or International Baccalaureate (IB) Exams AP Exam IB Exam AP and IB Exams I. Student Information Last Name First Name MI Grade High
iclef-2002 at Universities of Alicante and Jaen University of Alicante (Spain)
 iclef-2002 at Universities of Alicante and Jaen University of Alicante (Spain) ! Introduction! Passage Retrieval Systems! IR-n system! IR-n system at iclef-2002! Conclusions and Future works ! Introduction!
iclef-2002 at Universities of Alicante and Jaen University of Alicante (Spain) ! Introduction! Passage Retrieval Systems! IR-n system! IR-n system at iclef-2002! Conclusions and Future works ! Introduction!
PSA Y CRIBADO DEL CÁNCER DE PRÓSTATA: EVIDENCIAS CIENTÍFICAS ROBERTO LABAJO ROBLEDO MÉDICO DE FAMILIA C. S. SEGOVIA I SEGOVIA 2 NOVIEMBRE 2010
 PSA Y CRIBADO DEL CÁNCER DE PRÓSTATA: EVIDENCIAS CIENTÍFICAS ROBERTO LABAJO ROBLEDO MÉDICO DE FAMILIA C. S. SEGOVIA I SEGOVIA 2 NOVIEMBRE 2010 CRIBADO / SCREENING Detección en estadío precoz (preclínico
PSA Y CRIBADO DEL CÁNCER DE PRÓSTATA: EVIDENCIAS CIENTÍFICAS ROBERTO LABAJO ROBLEDO MÉDICO DE FAMILIA C. S. SEGOVIA I SEGOVIA 2 NOVIEMBRE 2010 CRIBADO / SCREENING Detección en estadío precoz (preclínico
Detección Marcadores Moleculares por PCR. Dr. Carlos G. Gonzalez Becuar
 Detección Marcadores Moleculares por PCR Dr. Carlos G. Gonzalez Becuar Virus de Papiloma Humano (VPH) - El CaCu ocupa los primeros lugares por muertes por Cancer en Mexico - Factor principal: VPH tipos
Detección Marcadores Moleculares por PCR Dr. Carlos G. Gonzalez Becuar Virus de Papiloma Humano (VPH) - El CaCu ocupa los primeros lugares por muertes por Cancer en Mexico - Factor principal: VPH tipos
Efectividad y seguridad de la artroplastia total discal en lumbalgia crónica. Revisión sistemática y metaanálisis
 Efectividad y seguridad de la artroplastia total discal en lumbalgia crónica. Revisión sistemática y metaanálisis Lumbar total disc replacement effectiveness and safety in chronic low back pain. Executive
Efectividad y seguridad de la artroplastia total discal en lumbalgia crónica. Revisión sistemática y metaanálisis Lumbar total disc replacement effectiveness and safety in chronic low back pain. Executive
ESTUDIO CLÍNICO CON IMPLANTES DE SUPERFICIE OXALIFE EN PACIENTES FUMADORES Y NO FUMADORES
 ESTUDIO CLÍNICO CON IMPLANTES DE SUPERFICIE OXALIFE EN PACIENTES FUMADORES Y NO FUMADORES OXALIFE IMPLANTS SURFACE IN SMOKING AND NO SMOKING PATIENTS: A CLINICAL STUDY Autor: Malbos Marisel Director: Ibáñez
ESTUDIO CLÍNICO CON IMPLANTES DE SUPERFICIE OXALIFE EN PACIENTES FUMADORES Y NO FUMADORES OXALIFE IMPLANTS SURFACE IN SMOKING AND NO SMOKING PATIENTS: A CLINICAL STUDY Autor: Malbos Marisel Director: Ibáñez
Detección del Ganglio Centinela en Patología Abdominal. Dra. C Balagué
 Detección del Ganglio Centinela en Patología Abdominal Dra. C Balagué DISEMINACIÓN LINFÁTICA Patrón ordenado, secuencial y predecible a través de los conductos linfáticos. GANGLIO CENTINELA Ganglios con
Detección del Ganglio Centinela en Patología Abdominal Dra. C Balagué DISEMINACIÓN LINFÁTICA Patrón ordenado, secuencial y predecible a través de los conductos linfáticos. GANGLIO CENTINELA Ganglios con
Consejería de Salud Agencia de Evaluación de Tecnologías Sanitarias de Andalucía
 CONSEJERÍA DE SALUD Coste-efectividad del palivizumab en la prevención de la hospitalización por virus respiratorio sincitial en prematuros de 32 a 35 semanas de gestación. Actualización del informe 5
CONSEJERÍA DE SALUD Coste-efectividad del palivizumab en la prevención de la hospitalización por virus respiratorio sincitial en prematuros de 32 a 35 semanas de gestación. Actualización del informe 5
Tratamiento de combinación en el melanoma irresecable o avanzado BRAF + Dra. Ainara Soria Rivas Servicio de Oncología Médica
 Tratamiento de combinación en el melanoma irresecable o avanzado BRAF + Dra. Ainara Soria Rivas Servicio de Oncología Médica EVOLUCIÓN DEL TRATAMIENTO DEL MELANOMA DACARBAZINA Combinaciones de quimioterapia
Tratamiento de combinación en el melanoma irresecable o avanzado BRAF + Dra. Ainara Soria Rivas Servicio de Oncología Médica EVOLUCIÓN DEL TRATAMIENTO DEL MELANOMA DACARBAZINA Combinaciones de quimioterapia
INSTITUTO POLITÉCNICO NACIONAL
 INSTITUTO POLITÉCNICO NACIONAL ESCUELA SUPERIOR DE TURISMO SECCIÓN DE ESTUDIOS DE POSGRADO E INVESTIGACIÓN MAESTRÍA EN ADMINISTRACIÓN E INNOVACIÓN DEL TURISMO ANÁLISIS DEL PROCESO DE GESTIÓN DEL TALENTO
INSTITUTO POLITÉCNICO NACIONAL ESCUELA SUPERIOR DE TURISMO SECCIÓN DE ESTUDIOS DE POSGRADO E INVESTIGACIÓN MAESTRÍA EN ADMINISTRACIÓN E INNOVACIÓN DEL TURISMO ANÁLISIS DEL PROCESO DE GESTIÓN DEL TALENTO
GUÍA DE PRÁCTICA CLÍNICA. Detección Precoz del Cáncer de Próstata. Dr. Pablo González Granda
 Dr. Pablo González Granda Año 2013 - Revisión: 0 Página 1 de 6 Documento de Base Early Detection of Prostate Cancer: American Urological Association (disponible en http://www.auanet.org/common/pdf/education/clinical-guidance/prostate-cancer-
Dr. Pablo González Granda Año 2013 - Revisión: 0 Página 1 de 6 Documento de Base Early Detection of Prostate Cancer: American Urological Association (disponible en http://www.auanet.org/common/pdf/education/clinical-guidance/prostate-cancer-
Efectividad de la automonitorización y autocontrol del tratamiento con anticoagulantes orales antagonistas de la vitamina K
 Efectividad de la automonitorización y autocontrol del tratamiento con anticoagulantes orales antagonistas de la vitamina K Revisión sistemática Effectiveness of patient self-testing and patient self-management
Efectividad de la automonitorización y autocontrol del tratamiento con anticoagulantes orales antagonistas de la vitamina K Revisión sistemática Effectiveness of patient self-testing and patient self-management
TEXAS DEPARTMENT OF STATE HEALTH SERVICES
 TEXAS DEPARTMENT OF STATE HEALTH SERVICES DAVID L. LAKEY, M.D. COMMISSIONER P.O. Box 149347 Austin, Texas 78714-9347 1-888-963-7111 TTY: 1-800-735-2989 www.dshs.state.tx.us August 15, 2013 Dear Birthing
TEXAS DEPARTMENT OF STATE HEALTH SERVICES DAVID L. LAKEY, M.D. COMMISSIONER P.O. Box 149347 Austin, Texas 78714-9347 1-888-963-7111 TTY: 1-800-735-2989 www.dshs.state.tx.us August 15, 2013 Dear Birthing
2014 15 Student Eligibility Verification Advanced Placement/International Baccalaureate Test Fee Program
 2014 15 Student Eligibility Verification Advanced Placement (AP) and/or International Baccalaureate (IB) Exams þ AP Exam IB Exam AP and IB Exams I. Student Information Last Name First Name MI Grade High
2014 15 Student Eligibility Verification Advanced Placement (AP) and/or International Baccalaureate (IB) Exams þ AP Exam IB Exam AP and IB Exams I. Student Information Last Name First Name MI Grade High
Creating your Single Sign-On Account for the PowerSchool Parent Portal
 Creating your Single Sign-On Account for the PowerSchool Parent Portal Welcome to the Parent Single Sign-On. What does that mean? Parent Single Sign-On offers a number of benefits, including access to
Creating your Single Sign-On Account for the PowerSchool Parent Portal Welcome to the Parent Single Sign-On. What does that mean? Parent Single Sign-On offers a number of benefits, including access to
PowerPoint Slides English Spanish Translation Breast Cancer Survivorshiop: Epidemiology Video Transcript
 PowerPoint Slides English Spanish Translation Breast Cancer Survivorshiop: Epidemiology Video Transcript Supervivencia al cáncer de mama: epidemiología Transcripción del video Professional Oncology Education
PowerPoint Slides English Spanish Translation Breast Cancer Survivorshiop: Epidemiology Video Transcript Supervivencia al cáncer de mama: epidemiología Transcripción del video Professional Oncology Education
Volatilidad: Noviembre 2010 Futuros Frijol de Soya
 Observaciones Junio 09, 2010 1. La volatilidad tiene una tendencia a aumentar de Junio a Julio. 2. Este reporte sugiere que se debería considerar la implementación de estrategias largas con opciones en
Observaciones Junio 09, 2010 1. La volatilidad tiene una tendencia a aumentar de Junio a Julio. 2. Este reporte sugiere que se debería considerar la implementación de estrategias largas con opciones en
IntesisBox PA-RC2-xxx-1 Panasonic compatibilities
 IntesisBox PA-RC2-xxx-1 Panasonic compatibilities In this document the compatible Panasonic models with the following IntesisBox RC2 interfaces are listed: / En éste documento se listan los modelos PANASONIC
IntesisBox PA-RC2-xxx-1 Panasonic compatibilities In this document the compatible Panasonic models with the following IntesisBox RC2 interfaces are listed: / En éste documento se listan los modelos PANASONIC
UNIT 9.- INTRODUCTION TO HYPOTHESIS TESTING.
 STATISTICAL METHODS FOR BUSINESS UNIT 9.- INTRODUCTION TO HYPOTHESIS TESTING. 9.1.- Basics of statistical hypothesis testing. 9.2.- Types of errors in hypothesis testing. 9.3.- Methodology and implementation
STATISTICAL METHODS FOR BUSINESS UNIT 9.- INTRODUCTION TO HYPOTHESIS TESTING. 9.1.- Basics of statistical hypothesis testing. 9.2.- Types of errors in hypothesis testing. 9.3.- Methodology and implementation
Efficacy, safety and efficiency of transcutaneous nerve stimulation for the treatment of chronic musculoskeletal pain.
 Eficacia, seguridad y coste-efectividad de la estimulación nerviosa eléctrica transcutánea (TENS) en el tratamiento del dolor musculoesquelético crónico Efficacy, safety and efficiency of transcutaneous
Eficacia, seguridad y coste-efectividad de la estimulación nerviosa eléctrica transcutánea (TENS) en el tratamiento del dolor musculoesquelético crónico Efficacy, safety and efficiency of transcutaneous
SAFETY ROAD SHOW 2015 Paul Teboul Co Chairman HST México
 SAFETY ROAD SHOW 2015 Paul Teboul Co Chairman HST México How did started the Safety Road Show? Airbus Helicopters and others manufacturers did invest since a long time in improving Aviation Safety. In
SAFETY ROAD SHOW 2015 Paul Teboul Co Chairman HST México How did started the Safety Road Show? Airbus Helicopters and others manufacturers did invest since a long time in improving Aviation Safety. In
ESTUDIO OBINDIAB GRUPO DE DIABETES SEMFYC
 ESTUDIO OBINDIAB GRUPO DE DIABETES SEMFYC METHODS CHARACTERISTICS OF THE PATIENTS Mean (SD) Range CI95% Age (years) 68,9+11,3 40-95 (67,8-70,0) HbA1c (%) 6,97+1,2 5,0-14,7 (6,9-7,1) Gender Male Female
ESTUDIO OBINDIAB GRUPO DE DIABETES SEMFYC METHODS CHARACTERISTICS OF THE PATIENTS Mean (SD) Range CI95% Age (years) 68,9+11,3 40-95 (67,8-70,0) HbA1c (%) 6,97+1,2 5,0-14,7 (6,9-7,1) Gender Male Female
Cancer Incidence in Latin America: An Overview
 Cancer Incidence in Latin America: An Overview Maria Paula Curado International Agency for Research on Cancer Lyon, France Cancer Incidence in 11% world population Introduction - Total population in 2006
Cancer Incidence in Latin America: An Overview Maria Paula Curado International Agency for Research on Cancer Lyon, France Cancer Incidence in 11% world population Introduction - Total population in 2006
IntesisBox MD-AC-xxx-yy AC indoor unit compatibilities
 IntesisBox MD-AC-xxx-yy AC indoor unit compatibilities In this document the compatible Midea Air conditioner indoor units models with the following IntesisBox AC interfaces are described: / En éste documento
IntesisBox MD-AC-xxx-yy AC indoor unit compatibilities In this document the compatible Midea Air conditioner indoor units models with the following IntesisBox AC interfaces are described: / En éste documento
PowerPoint Slides English Spanish Translation ICO2: Screening for Cancer Video Transcript
 PowerPoint Slides English Spanish Translation ICO2: Screening for Cancer Video Transcript ICO2: Pruebas de detección de cáncer Transcripción del video Professional Oncology Education ICO2: Screening for
PowerPoint Slides English Spanish Translation ICO2: Screening for Cancer Video Transcript ICO2: Pruebas de detección de cáncer Transcripción del video Professional Oncology Education ICO2: Screening for
English Literacy Success Team, e3 Civic High October 30, 2014
 English Literacy Success Team, e3 Civic High October 30, 2014 What is the English Language Success Team? The purpose of our committee is to provide an open conversation between parents, students, and teachers
English Literacy Success Team, e3 Civic High October 30, 2014 What is the English Language Success Team? The purpose of our committee is to provide an open conversation between parents, students, and teachers
PETER T. KATZMARZYK, PhD, FACSM
 PETER T. KATZMARZYK, PhD, FACSM Dr. Katzmarzyk actualmente es profesor y Director Ejecutivo del Centro de Investigación Biomédico de Pennington en Baton Rouge, Louisiana, Ha sido presidente experto en
PETER T. KATZMARZYK, PhD, FACSM Dr. Katzmarzyk actualmente es profesor y Director Ejecutivo del Centro de Investigación Biomédico de Pennington en Baton Rouge, Louisiana, Ha sido presidente experto en
Cáncer de mama metastásico ER-/HER2+ y resistencia precoz a la terapia con trastuzumab
 Cáncer de mama metastásico ER-/HER2+ y resistencia precoz a la terapia con trastuzumab Eva M Ciruelos Gil Hospital Universitario 12 de Octubre, Madrid Caso clínico Paciente con cáncer de mama HER2+ y recidiva
Cáncer de mama metastásico ER-/HER2+ y resistencia precoz a la terapia con trastuzumab Eva M Ciruelos Gil Hospital Universitario 12 de Octubre, Madrid Caso clínico Paciente con cáncer de mama HER2+ y recidiva
PEDRO SÁNCHEZ ROVIRA COMPLEJO HOSPITALARIO DE JAÉN
 PEDRO SÁNCHEZ ROVIRA COMPLEJO HOSPITALARIO DE JAÉN Maintenance therapy is the treatment of cancer with medication, typically following an initial round of treatment. Maintenance treatment may include chemotherapy,
PEDRO SÁNCHEZ ROVIRA COMPLEJO HOSPITALARIO DE JAÉN Maintenance therapy is the treatment of cancer with medication, typically following an initial round of treatment. Maintenance treatment may include chemotherapy,
La hipertensión arterial en el 2013: las nuevas guías de la ESH/ESC. Alejandro de la Sierra Hospital Mutua Terrassa
 La hipertensión arterial en el 2013: las nuevas guías de la ESH/ESC Alejandro de la Sierra Hospital Mutua Terrassa Universidad de Barcelona J Hypertens 2013; 31: 1281-1357 Eur Heart J 2013; 34: 2159-2219
La hipertensión arterial en el 2013: las nuevas guías de la ESH/ESC Alejandro de la Sierra Hospital Mutua Terrassa Universidad de Barcelona J Hypertens 2013; 31: 1281-1357 Eur Heart J 2013; 34: 2159-2219
Benchmarking de Transporte México. Mayo 2013
 Benchmarking de Transporte México Mayo 2013 1 Benchmarking de Transporte México (Primario) Objetivo: El desarrollo de medidas comparativas (benchmarking) de aspectos cualitativos/cuantitativos del transporte
Benchmarking de Transporte México Mayo 2013 1 Benchmarking de Transporte México (Primario) Objetivo: El desarrollo de medidas comparativas (benchmarking) de aspectos cualitativos/cuantitativos del transporte
Combinaciones Quimioterápicas en Hemopatías Malignas. Linfoma de Hodgkin
 Combinaciones Quimioterápicas en Hemopatías Malignas. Linfoma de Hodgkin Anna Sureda Servei d Hematologia Institut Catala d Oncologia Hospital Duran i Reynals Barcelona Tratamiento Antineoplásico en Hematología:
Combinaciones Quimioterápicas en Hemopatías Malignas. Linfoma de Hodgkin Anna Sureda Servei d Hematologia Institut Catala d Oncologia Hospital Duran i Reynals Barcelona Tratamiento Antineoplásico en Hematología:
Epidemiología del Cáncer. Incidencia. Mortalidad
 Biología Molecular del Cáncer y Nuevas Dianas Terapéuticas Epidemiología del Cáncer. Incidencia. Mortalidad Dr. Ignacio Blanco Director, Programa de Consell Genètic en Càncer Institut Català d Oncología
Biología Molecular del Cáncer y Nuevas Dianas Terapéuticas Epidemiología del Cáncer. Incidencia. Mortalidad Dr. Ignacio Blanco Director, Programa de Consell Genètic en Càncer Institut Català d Oncología
Prolaris (CCP Score) ; Test pronóstico en Cáncer de Próstata Dr J Rubio Briones Instituto Valenciano de Oncología
 Prolaris (CCP Score) ; Test pronóstico en Cáncer de Próstata Dr J Rubio Briones Instituto Valenciano de Oncología Screening en Cáncer de próstata A favor: 3ª causa de muerte en hombres por tumor Disminuye
Prolaris (CCP Score) ; Test pronóstico en Cáncer de Próstata Dr J Rubio Briones Instituto Valenciano de Oncología Screening en Cáncer de próstata A favor: 3ª causa de muerte en hombres por tumor Disminuye
Tratamiento de los pacientes con recidiva de ETV a pesar de tratamiento anticoagulante oral. Dr Lobo S de Neumología. Hospital Txagorritxu.
 Tratamien de los pacientes con recidiva de ETV a pesar de tratamien anticoagulante oral Dr Lobo S de Neumología. Hospital Txagorritxu. Viria anticoagulación oral con antivitamas K (AVK) a largo plazo 4-7%
Tratamien de los pacientes con recidiva de ETV a pesar de tratamien anticoagulante oral Dr Lobo S de Neumología. Hospital Txagorritxu. Viria anticoagulación oral con antivitamas K (AVK) a largo plazo 4-7%
Are You Eligible for a FREE Mammogram? Es Usted Elegible Para una Mamografía GRATIS? C OLUMBIA U NIVERSITY B REAST C ANCER S CREENING P ARTNERSHIP
 C OLUMBIA U NIVERSITY B REAST C ANCER S CREENING P ARTNERSHIP Are You Eligible for a FREE Mammogram? Es Usted Elegible Para una Mamografía GRATIS? Columbia Presbyterian Center NewYork-Presbyterian Hospital
C OLUMBIA U NIVERSITY B REAST C ANCER S CREENING P ARTNERSHIP Are You Eligible for a FREE Mammogram? Es Usted Elegible Para una Mamografía GRATIS? Columbia Presbyterian Center NewYork-Presbyterian Hospital
Personal Dosimetry Statistics and Specifics of Low Dose Evaluation
 Personal Dosimetry Statistics and Specifics of Low Dose Evaluation Comisión Chilena de Energía Nuclear Ricardo Avila, Roberto Gómez, Personal Dosimetry Laboratory and Carlos Oyarzún, Metrology of Ionizing
Personal Dosimetry Statistics and Specifics of Low Dose Evaluation Comisión Chilena de Energía Nuclear Ricardo Avila, Roberto Gómez, Personal Dosimetry Laboratory and Carlos Oyarzún, Metrology of Ionizing
Dispositivo totalmente implantable Esteem en el tratamiento de la hipoacusia neurosensorial
 Dispositivo totalmente implantable Esteem en el tratamiento de la hipoacusia neurosensorial Revisión sistemática Esteem totally implantable hearing device for treatment of sensorineural hearing loss. Systematic
Dispositivo totalmente implantable Esteem en el tratamiento de la hipoacusia neurosensorial Revisión sistemática Esteem totally implantable hearing device for treatment of sensorineural hearing loss. Systematic
Cáncer de próstata localizado. Opciones de tratamiento según grupos pronósticos. Fco José Anglada Curado Urología Hospital Reina Sofía
 Cáncer de próstata localizado. Opciones de tratamiento según grupos pronósticos Fco José Anglada Curado Urología Hospital Reina Sofía Riesgo 15-18% de padecer la enfermedad durante la vida 90% localizados
Cáncer de próstata localizado. Opciones de tratamiento según grupos pronósticos Fco José Anglada Curado Urología Hospital Reina Sofía Riesgo 15-18% de padecer la enfermedad durante la vida 90% localizados
MI ENTORNO DE TRABAJO ES EL MUNDO
 MI ENTORNO DE TRABAJO ES EL MUNDO EL IDIOMA INTERNACIONAL ES EL INGLÉS EL CONVENIO STCW95 LO ESTIPULA COMO EL IDIOMA DE COMUNICACIÓN DE LOS OFICIALES MARLINS ES LA ÚNICA HERRAMIENTA PARA ACREDITAR EL NIVEL
MI ENTORNO DE TRABAJO ES EL MUNDO EL IDIOMA INTERNACIONAL ES EL INGLÉS EL CONVENIO STCW95 LO ESTIPULA COMO EL IDIOMA DE COMUNICACIÓN DE LOS OFICIALES MARLINS ES LA ÚNICA HERRAMIENTA PARA ACREDITAR EL NIVEL
Barclaycard Center Identidad Visual / Visual Identity Uso de la marca como referencia / Use of the brand as reference
 Barclaycard Center Identidad Visual / Visual Identity Uso de la marca como referencia / Use of the brand as reference Julio 2014 / July 2014 Contenidos / Contents 02 Eventos / Eventos 3 14 15 16 Aplicacion
Barclaycard Center Identidad Visual / Visual Identity Uso de la marca como referencia / Use of the brand as reference Julio 2014 / July 2014 Contenidos / Contents 02 Eventos / Eventos 3 14 15 16 Aplicacion
Cribado prenatal no invasivo
 Cribado prenatal no invasivo Detección T13, T18, T21, monosomía X y sexo fetal en sangre materna. Dra. Blanca Bermejo Barrera, Directora Desarrollo Área Molecular Evolución del cribado prenatal 1960 s
Cribado prenatal no invasivo Detección T13, T18, T21, monosomía X y sexo fetal en sangre materna. Dra. Blanca Bermejo Barrera, Directora Desarrollo Área Molecular Evolución del cribado prenatal 1960 s
Inmunoterapia activa para el cáncer de mama
 Inmunoterapia activa para el cáncer de mama Revisión sistemática Informe de síntesis de tecnología emergente Active immunotherapy for breast cancer. Systematic review Executive summary INFORMES DE EVALUACIÓN
Inmunoterapia activa para el cáncer de mama Revisión sistemática Informe de síntesis de tecnología emergente Active immunotherapy for breast cancer. Systematic review Executive summary INFORMES DE EVALUACIÓN
Detección precoz del cáncer de mama: cribados organizados y oportunísticos
 Detección precoz del cáncer de mama: cribados organizados y oportunísticos Francisco Pérez Riquelme Médico Especialista en Salud Pública y Medicina Preventiva Coordinador del Programa de Prevención del
Detección precoz del cáncer de mama: cribados organizados y oportunísticos Francisco Pérez Riquelme Médico Especialista en Salud Pública y Medicina Preventiva Coordinador del Programa de Prevención del
Resumen. Abstract CÁNCER: UN FORMIDABLE RETO. Gaceta Médica de México. 2014;150 Suppl 2:140-4
 Gaceta Médica de México. 2014;150 Suppl 2:140-4 CÁNCER: UN FORMIDABLE RETO Comparación entre vigilancia y radioterapia adyuvante o de salvamento después de una prostatectomía radical para cáncer de próstata
Gaceta Médica de México. 2014;150 Suppl 2:140-4 CÁNCER: UN FORMIDABLE RETO Comparación entre vigilancia y radioterapia adyuvante o de salvamento después de una prostatectomía radical para cáncer de próstata
What is family health history?
 Family Health History Project Pre-Survey What is family health history? Family health history is information about diseases that run in your family, as well as the eating habits, activities, and environments
Family Health History Project Pre-Survey What is family health history? Family health history is information about diseases that run in your family, as well as the eating habits, activities, and environments
Determinación de la Incidencia de Cáncer e Hiperplasia Benigna de Próstata, Mediante la Cuantificación
 DETERMINACIÓN DE LA INCIDENCIA DE CÁNCER E HIPERPLASIA BENIGNA DE PRÓSTATA, MEDIANTE LA CUANTIFICACIÓN DE PSA TOTAL Y PSA LIBRE, EN PACIENTES QUE ASISTIERON AL INSTITUTO SELADIS DURANTE EL PERIÓDO PELAEZ
DETERMINACIÓN DE LA INCIDENCIA DE CÁNCER E HIPERPLASIA BENIGNA DE PRÓSTATA, MEDIANTE LA CUANTIFICACIÓN DE PSA TOTAL Y PSA LIBRE, EN PACIENTES QUE ASISTIERON AL INSTITUTO SELADIS DURANTE EL PERIÓDO PELAEZ
DUAL IMMERSION PROGRAM INFORMATION PRESCHOOL PRESENTATION SEPTEMBER 10, 2014 6:30 P.M.
 DUAL IMMERSION PROGRAM INFORMATION PRESCHOOL PRESENTATION SEPTEMBER 10, 2014 6:30 P.M. Presented by Dr. Norma R. Delgado, Director of Curriculum & Instruction 1 The United States Government has identified
DUAL IMMERSION PROGRAM INFORMATION PRESCHOOL PRESENTATION SEPTEMBER 10, 2014 6:30 P.M. Presented by Dr. Norma R. Delgado, Director of Curriculum & Instruction 1 The United States Government has identified
EN United in diversity EN A8-0204/83. Amendment
 27.6.2018 A8-0204/83 83 Recital 2 (2) So far, and unless otherwise provided for in national law, the rules on access to the occupation of road transport operator do not apply to undertakings engaged in
27.6.2018 A8-0204/83 83 Recital 2 (2) So far, and unless otherwise provided for in national law, the rules on access to the occupation of road transport operator do not apply to undertakings engaged in
QUE ES EL CANCER DE MAMA PDF
 QUE ES EL CANCER DE MAMA PDF ==> Download: QUE ES EL CANCER DE MAMA PDF QUE ES EL CANCER DE MAMA PDF - Are you searching for Que Es El Cancer De Mama Books? Now, you will be happy that at this time Que
QUE ES EL CANCER DE MAMA PDF ==> Download: QUE ES EL CANCER DE MAMA PDF QUE ES EL CANCER DE MAMA PDF - Are you searching for Que Es El Cancer De Mama Books? Now, you will be happy that at this time Que
SOCIEDAD DE MASTOLOGIA 6 DE MAYO 2010 ONCOTYPE DX. Dr. Francisco Domínguez
 SOCIEDAD DE MASTOLOGIA 6 DE MAYO 2010 ONCOTYPE DX Dr. Francisco Domínguez Departamento de Cirugía Oncológica Facultad de Medicina Pontificia Universidad Católica de Chile Conceptos generales Diagnóstico
SOCIEDAD DE MASTOLOGIA 6 DE MAYO 2010 ONCOTYPE DX Dr. Francisco Domínguez Departamento de Cirugía Oncológica Facultad de Medicina Pontificia Universidad Católica de Chile Conceptos generales Diagnóstico
QUÈ PASSA AMB LA RDT DE CADENES GANGLIONARS. Dra. Arancha Eraso Servei d Oncologia Radioteràpia
 QUÈ PASSA AMB LA RDT DE CADENES GANGLIONARS. Dra. Arancha Eraso Servei d Oncologia Radioteràpia Pre- ACOSOG Z0011 y MA.20 SUPRA I II Paciente cn1 o GC positivo IRRADIACIÓN CADENAS IRRADIACIÓN SUPRACLAVICULAR
QUÈ PASSA AMB LA RDT DE CADENES GANGLIONARS. Dra. Arancha Eraso Servei d Oncologia Radioteràpia Pre- ACOSOG Z0011 y MA.20 SUPRA I II Paciente cn1 o GC positivo IRRADIACIÓN CADENAS IRRADIACIÓN SUPRACLAVICULAR
Fall Notice to Beneficiaries Enrolled in Low Performing Plans Information for SHIPs
 Fall Notice to Beneficiaries Enrolled in Low Performing Plans Information for SHIPs CMS is sending a notice to Medicare beneficiaries currently enrolled in consistently low performing plans. This notice
Fall Notice to Beneficiaries Enrolled in Low Performing Plans Information for SHIPs CMS is sending a notice to Medicare beneficiaries currently enrolled in consistently low performing plans. This notice

 Full version is >>> HERE
Full version is >>> HERE